Cell Culture Media for Vaccine Production
Cell culture media for vaccine production are optimized nutrient systems used to expand production cell lines and support controlled viral propagation in research and manufacturing workflows. For buyers, the practical questions are consistency, suitability for the target cell line, documentation depth, supply continuity, and total landed cost. CellCultureMedia supplies media, sera, supplements, and process reagents for Vero, MDCK, BHK-21, PK-15, CEF, and related platforms, including serum-free and animal-derived component-free options. We support R&D screening, scale-up, and routine production sourcing with responsive quotation, lot planning, and free worldwide shipping where it fits the order profile.
What this category/application covers
Vaccine production media support the growth and maintenance of host cell lines used for viral antigen generation, process development, seed train work, and upstream manufacturing. This application includes chemically defined, serum-free, low-protein, and classical serum-supplemented systems, depending on the cell line, process history, and regulatory expectations for the finished program.
Typical users include biotech manufacturers, contract production teams, academic core facilities, and translational R&D groups that need dependable media supply without adding complexity to qualification. CellCultureMedia can support small-volume feasibility studies as well as repeated batch purchasing for larger campaigns. Related upstream materials are organized under bioprocess media for teams comparing broader production and scale-up inputs.
Common products and formulations
- Vero cell media: formulations for adherent or microcarrier-based workflows, including serum-reduced and serum-free options for viral vaccine process development.
- MDCK cell media: nutrient systems for suspension or adherent influenza-related workflows, with attention to growth density, stability, and downstream compatibility.
- BHK-21 media: media for high-density growth and viral antigen production studies, available in classical and lower animal-origin formats.
- PK-15 and porcine cell media: options for swine-associated research and production cell lines where consistency and passage stability matter.
- CEF and avian cell media: media and supplements for primary or established avian workflows, supplied with practical documentation for lab qualification.
- Supplements and reagents: sera, buffers, antibiotics for research use, detachment reagents, cryopreservation solutions, and process support materials from cell culture reagents.
For buyers moving away from serum, serum-free media can help reduce lot-to-lot variability and simplify raw material review.
How to choose
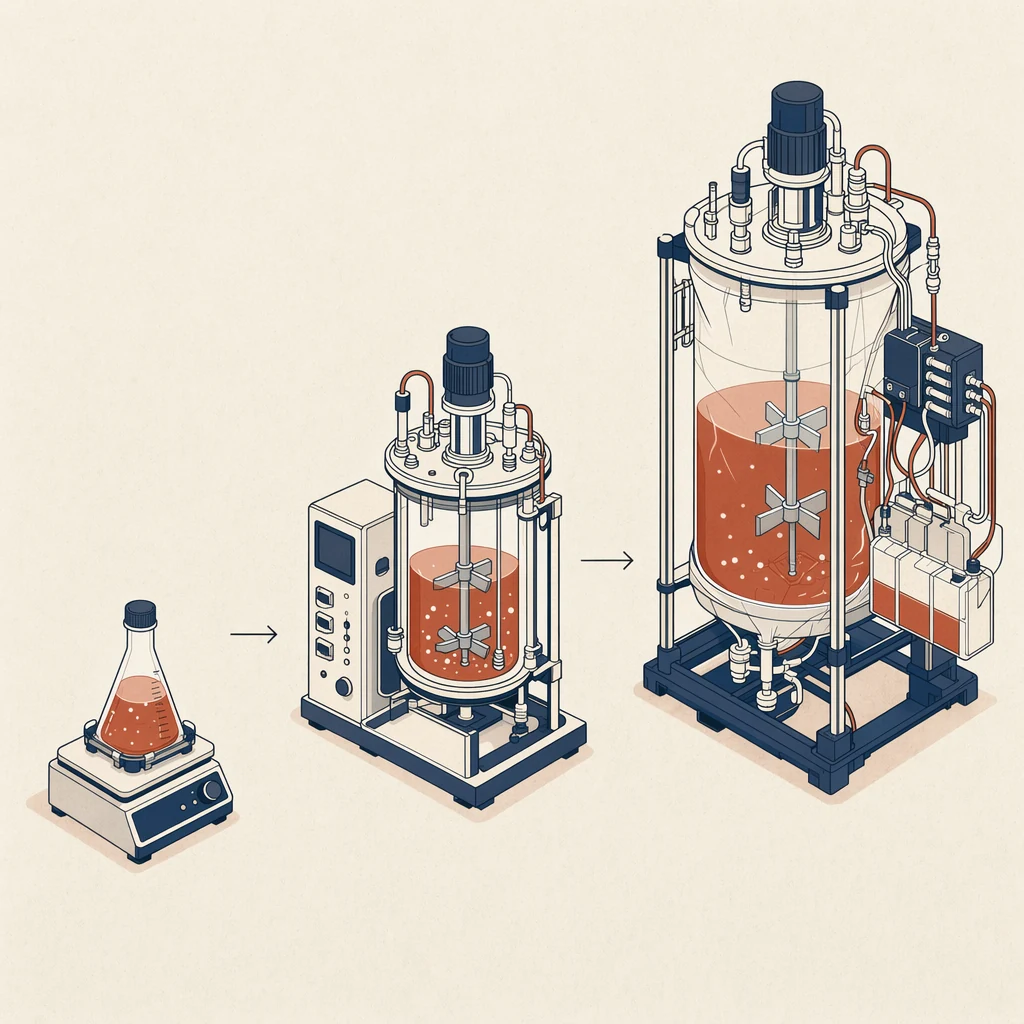
Start with the production cell line, growth mode, and scale. A medium that works in a static flask may need optimization before microcarrier, roller bottle, wave bag, or stirred-tank use. Confirm whether your process depends on serum, hydrolysates, attachment factors, or adaptation steps, because changing those variables can affect cell density, morphology, viral yield, harvest timing, and downstream clarification.
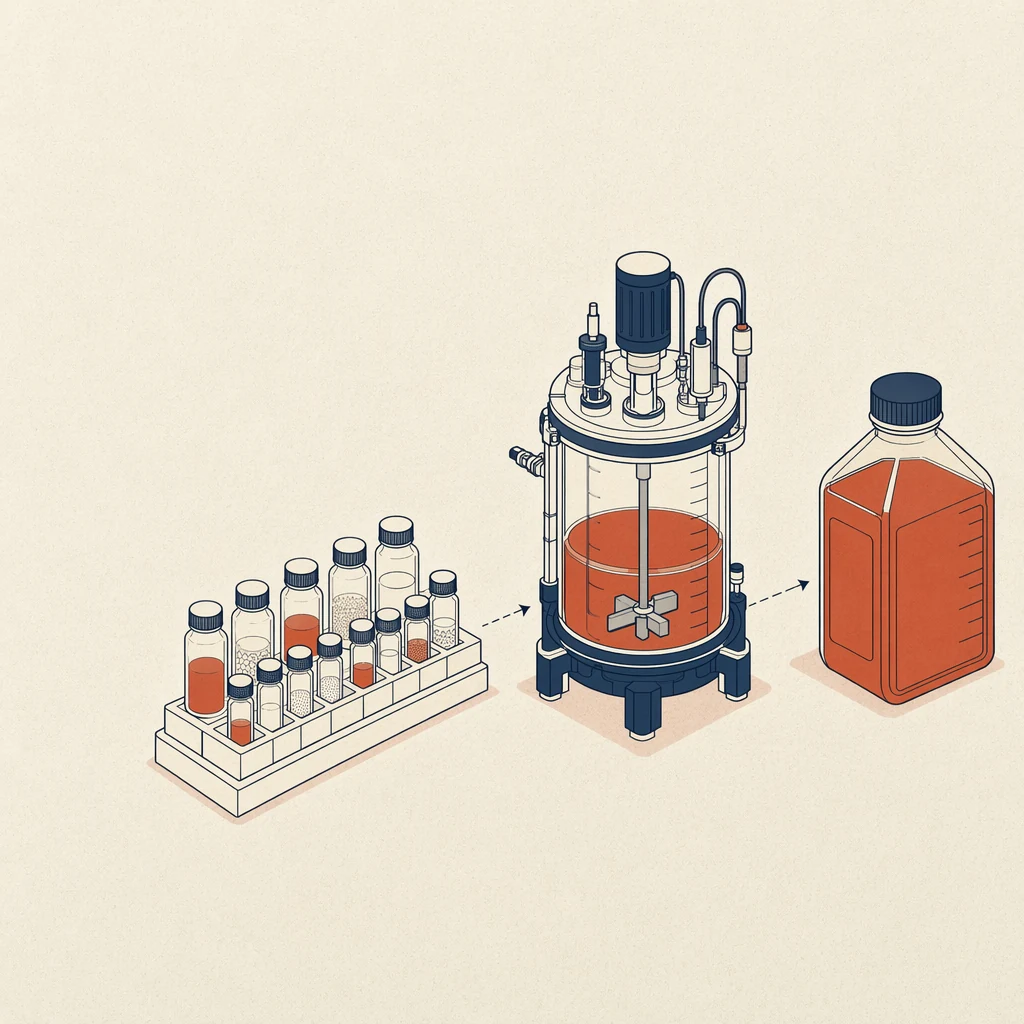
Next, define purchasing and technical criteria together. Buyers should compare pack size, lead time, minimum order quantity, shelf life, storage temperature, documentation, and freight conditions. Scientists should compare osmolality, pH range, protein content, animal-origin status, growth kinetics, and compatibility with transfection, infection, or harvest steps. When a catalog product is close but not exact, custom media services can align formulation, packaging, and testing with your internal specification.
Quality and documentation
For production media, quality review is not limited to a product name. Buyers typically need a certificate of analysis, safety data sheet, lot traceability, sterility statement, endotoxin data where applicable, origin information for animal-derived components, and change-control communication. CellCultureMedia works to provide clear documentation packages so sourcing teams can evaluate materials before purchase and keep records aligned with internal quality systems.
We recommend qualifying at least two lots when possible, especially for serum-containing or complex formulations. For scale-up, ask about lot reservation, multi-batch planning, and packaging that reduces handling risk. Our quality page outlines documentation practices, product handling expectations, and the type of technical files commonly available for media, sera, and reagents.
Why work with CellCultureMedia
- Application-aware sourcing: we understand the differences between Vero, MDCK, BHK-21, PK-15, CEF, and other production cell platforms.
- Flexible supply: source research packs, pilot volumes, and repeated production lots through one international supplier.
- Serum-free and ADCF support: compare formulations that reduce animal-origin exposure while preserving process practicality.
- Documentation focus: request COA, SDS, origin statements, and lot information during quotation instead of after delivery.
- Free worldwide shipping framing: we help teams evaluate delivered cost, lane feasibility, and temperature-control needs before order placement.
To move from specification to pricing, send your target cell line, format, annual volume, release documentation needs, and destination country through request a quote. Our team will respond with suitable catalog or custom options, lead time, free worldwide shipping framing, and available technical files.
Recommended Categories
For vaccine production workflows, customers most often need media and reagents from these categories. Each ships free worldwide.
Common questions about vaccine production
Do you supply media for both adherent and suspension production cell lines?
Can you provide serum-free or animal-derived component-free options?
What documents can be supplied with vaccine production media?
How should we evaluate a new medium before scale-up?
What information speeds up quotation and shipping review?
Discuss your vaccine production requirements
Tell us about your cell line, target scale, and regulatory context. We will recommend specific products, share documentation, and quote pricing.
Request a Quote